Author: Clinical Experts Group, Hamilton Medical
Date of first publication: 30.08.2017

Measurement on Hamilton Medical ventilators
Driving pressure = Plateau pressure ‑ Total PEEP
On a Hamilton Medical ventilator you can measure driving pressure in any ventilation mode, provided the patient is relaxed and making no inspiratory or expiratory effort, and there is no leakage from the ventilation circuit.
All you have to do is perform an end‑inspiratory hold maneuver and then an end‑expiratory hold for 3‑5 seconds to measure the plateau pressure and total PEEP respectively.
So driving pressure = inspiratory hold pressure (plateau pressure) ‑ expiratory hold pressure (total PEEP).
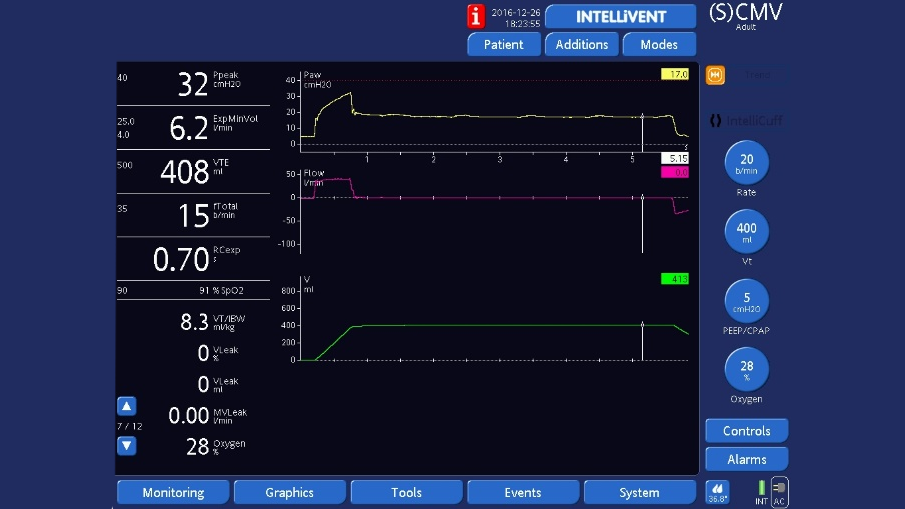
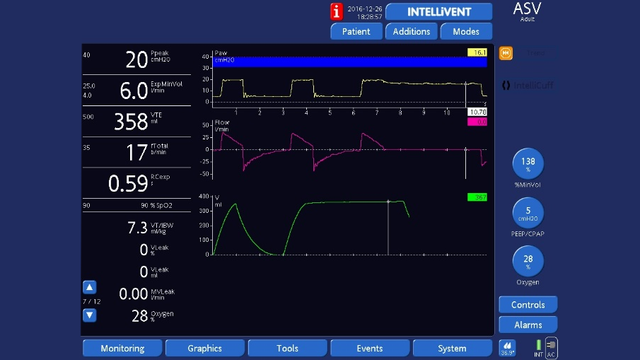
End‑inspiratory hold
End‑inspiratory hold: Press the Tools button to open the Tools ‑> Hold window and select Insp hold, wait for 3 to 5 seconds, then select Insp hold or press the P&T knob again to deactivate the hold maneuver and close the window. The waveforms will freeze so you can check that plateau pressure is stable, then measure pressure at the end of the plateau using the cursor.

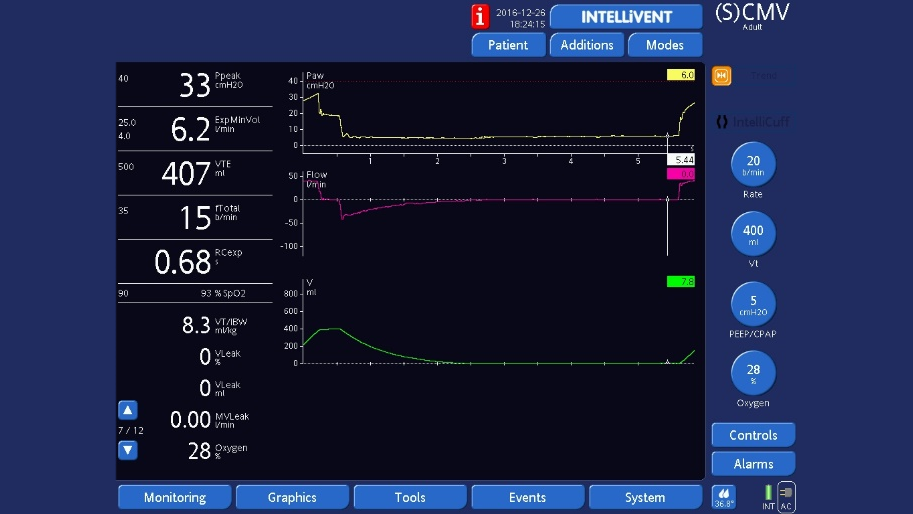
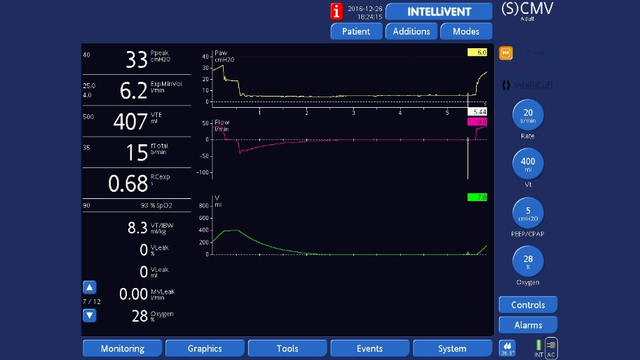
End‑expiratory hold
End‑expiratory hold: As for end‑inspiratory hold, but select Exp hold. Check that plateau pressure is stable, then measure pressure at the end of the plateau using the cursor.
Full citations below: (
Footnotes
References
- 1. Amato MB, Meade MO, Slutsky AS, et al. Driving pressure and survival in the acute respiratory distress syndrome. N Engl J Med. 2015;372(8):747‑755. doi:10.1056/NEJMsa1410639
- 2. Laffey JG, Bellani G, Pham T, et al. Potentially modifiable factors contributing to outcome from acute respiratory distress syndrome: the LUNG SAFE study [published correction appears in Intensive Care Med. 2017 Nov 14;:]. Intensive Care Med. 2016;42(12):1865‑1876. doi:10.1007/s00134‑016‑4571‑5
- 3. Guérin C, Papazian L, Reignier J, et al. Effect of driving pressure on mortality in ARDS patients during lung protective mechanical ventilation in two randomized controlled trials. Crit Care. 2016;20(1):384. Published 2016 Nov 29. doi:10.1186/s13054‑016‑1556‑2
- 4. Chiumello D, Carlesso E, Brioni M, Cressoni M. Airway driving pressure and lung stress in ARDS patients. Crit Care. 2016;20:276. Published 2016 Aug 22. doi:10.1186/s13054‑016‑1446‑7
- 5. Baedorf Kassis E, Loring SH, Talmor D. Mortality and pulmonary mechanics in relation to respiratory system and transpulmonary driving pressures in ARDS. Intensive Care Med. 2016;42(8):1206‑1213. doi:10.1007/s00134‑016‑4403‑7
- 6. Neto AS, Hemmes SN, Barbas CS, et al. Association between driving pressure and development of postoperative pulmonary complications in patients undergoing mechanical ventilation for general anaesthesia: a meta‑analysis of individual patient data [published correction appears in Lancet Respir Med. 2016 Jun;4(6):e34]. Lancet Respir Med. 2016;4(4):272‑280. doi:10.1016/S2213‑2600(16)00057‑6