Autor: Bernhard Schmitt; Mirko Belliato, MD (Pavia, Italy)
Datum: 29.11.2022
Last change: 29.11.2022
English additions made in German version
NIPPV mit Beatmungsgeräten von Hamilton Medical
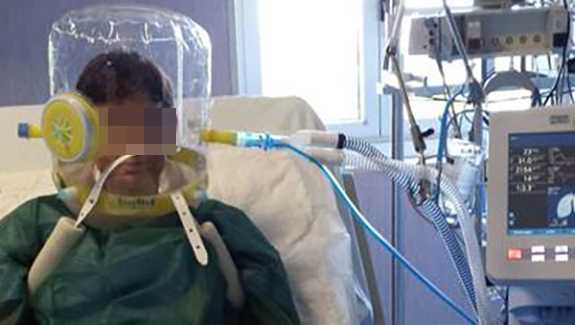
Die turbinenbetriebenen Beatmungsgeräte können höhere kontinuierliche Flowraten verabreichen und die Luftzufuhr wird mit HEPA‑gefilterter Raumluft mit Umgebungsfeuchtigkeit gespeist. Im Folgenden finden Sie eine schrittweise Anleitung zur Verwendung eines Helms für die NIPPV‑Beatmungstherapie bei erwachsenen COVID‑19‑Patienten mit einem Beatmungsgerät von Hamilton Medical.
HINWEIS! Beatmungsgeräte von Hamilton Medical dürfen nicht für die CPAP‑Therapie mit Helm verwendet werden.Stattdessen wird empfohlen, auf beiden Seiten des Helms ein ventilfreies System mit kontinuierlichem Flow zu verwenden (
Schritt 1: Konfiguration und Vorbereitung
- Verwenden Sie ein Beatmungsschlauchsystem mit zwei Schenkeln und niedriger Compliance und setzen Sie nach Möglichkeit aktive Befeuchtung ein (kann zu erheblicher Kondensation im Helm führen). Verwenden Sie kein koaxiales Schlauchsystem!
- Setzen Sie am Inspirations‑ und Exspirationsanschluss des Beatmungsgerätes einen Bakterien‑/Virenfilter ein.
- Aktivieren Sie den Hauptstrom‑ oder Nebenstrom‑CO2‑Sensor (sofern verfügbar) und versehen Sie ihn mit den erforderlichen Atemwegsadaptern.
- Führen Sie alle erforderlichen Überprüfungen vor Inbetriebnahme durch.
- Schließen Sie den Flow‑Sensor direkt an einem der Helmanschlüsse mit einem Konnektor mit 22 mm ID oder dem Kalibrationsadapter an.
- Verschließen Sie den zweiten Anschluss am Helm mit einer Kappe.
Schritt 2: Modusauswahl und Alarmeinstellungen
- Wählen Sie den NIV‑Modus am Beatmungsgerät aus (sofern verfügbar).
- Wenn keine NIV‑Option installiert ist, können Sie den Modus PCV+ oder PCMV wählen.
- Passen Sie die Alarmgrenzwerte so an, dass Fehlalarme vermieden werden.

Modusparameter
ACHTUNG! Zwei beatmete Kompartimente in Folge = Helm + Lungen.
| Druckrampe | Auf die höchstmögliche Geschwindigkeit eingestellt |
| PEEP | PEEP‑Zielwert + 30‑50 % Minimaler PEEP-Wert = 10 mbar für einen steiferen Helm |
| Psupport | Psupport‑Zielwert + 30‑50 % Minimaler Psupport-Wert = 12 mbar |
| Trigger für die Inspiration | Mit 2 l/min beginnen und möglichst niedrig halten |
| ETS | Mit ETS‑Standardeinstellung von 25 % beginnen, auf Asynchronien bei der Einleitung der Exspiration überwachen und entsprechend anpassen |
| Ti max | Auf 1,5 s einstellen, um eine späte Einleitung der Exspiration zu vermeiden |
| Sauerstoff | Mit einer Sauerstoffeinstellung von 60 % beginnen und auf Basis des SpO2‑Werts titrieren Hinweis: Bei einer einzigen Gasquelle (100 % Sauerstoff) kann die Peakflow-Kapazität begrenzt sein. |
Schritt 4: Monitoring
| Tidalvolumen | Zwischen 1.000 and 1.500 ml Hinweis: ~ 50‑75 % des verabreichten Vt wird an den Helm verteilt! |
| ExspMinVol | > 25 l/min für eine ausreichende CO2‑Auswaschung Die Wirksamkeit kann durch PCO2-Monitoring im Helm überwacht werden – siehe Tipps und Tricks unten. |
Tipps und Tricks
- Messen Sie den CO2‑Partialdruck im Helm (PCO2h) in einem „ruhigen“ Bereich des Helms (platzieren Sie z. B. den Sensor direkt über dem aufgeblasenen Kragen), um eine CO2‑Rückatmung zu erkennen. Verwenden Sie den Hauptstrom‑ oder Nebenstrom‑CO2‑Sensor des Beatmungsgerätes oder des Monitoringsystems. Der PCO2h‑Wert sollte 5 mmHg/0,6 kPa nicht übersteigen.
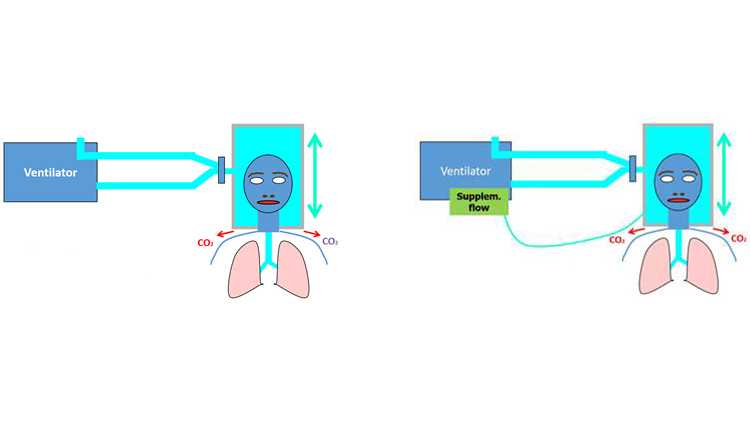
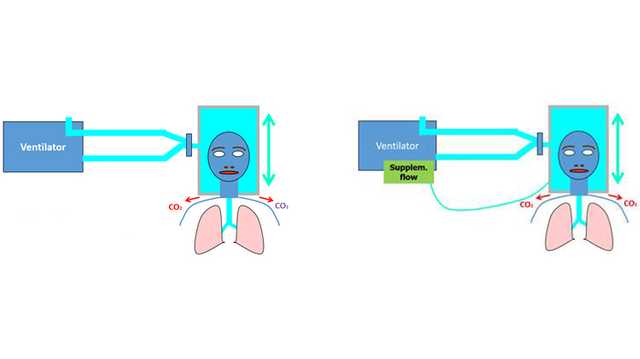
- Wenn eine CO2‑Rückatmung vermutet wird, führen Sie über den Anschluss für die Nahrungsaufnahme/den Zusatzanschluss am Helm einen ergänzenden Flow von > 10 l/min zu.
- Erhöhen Sie den Druckaufbau, indem Sie die Tubuskompensation (TRC) aktivieren (100 %).
(Bildmaterial freundlicherweise zur Verfügung gestellt von Dr. Mirko Belliato, Policlinico San Matteo Pavia Fondazione IRCCS s.c. Anestesia e Rianimazione II Pavia, Italien.)
Warum für NIPPV einen Helm verwenden?
Wird Sauerstoff über einen Nasenkatheter, eine Maske oder nichtinvasive Beatmung (NIV) verabreicht, entweicht eine erhebliche Menge ausgeatmete Luft in die Umgebung. Das kann die Verteilung von Viren und damit das Risiko einer nosokomialen Infektion erhöhen (
Es besteht die Hoffnung, dass die Beatmung mit Helm zur Verminderung des Risikos für eine nosokomiale Infektion beitragen kann: In diesem Fall wird anstelle der Gesichtsmaske ein Helm für die Verabreichung der nichtinvasiven Beatmung verwendet. Ein Vergleich der NIPPV‑Beatmung mit Helm mit der NIPPV‑Beatmung mit Gesichtsmaske ergab in einer simulierten Umgebung mit einem Intensivbeatmungsgerät, einem Schlauchsystem mit zwei Schenkeln und einem Filter am Exspirationsanschluss des Beatmungsgerätes, dass die Leckage von ausgeatmeter Luft bei der Beatmung mit Helm vernachlässigbar war (
Dennoch mahnen einige Experten beim Einsatz von Helmen zur Vorsicht. In den Richtlinien der ESICM werden sie als ansprechende Option beschrieben, da sie nachweislich die Verteilung von ausgeatmeter Luft reduzieren. Die Autoren betonen aber, dass sie nicht sicher sind, was die Sicherheit und Wirksamkeit von Helmen bei COVID‑19‑Patienten angeht. Daher konnten sie keine Empfehlung zur Verwendung der NIPPV‑Beatmung mit Helm gegenüber der NIPPV‑Beatmung mit Maske aussprechen (
Mein Beatmungsgerät von Hamilton Medical erlaubt keine Flow‑By‑Konfiguration...
Aufgrund des weit höheren Volumens (immer höher als das Tidalvolumen) ähnelt ein Helm einer halbgeschlossenen Umgebung wie einem geschlossenen Raum mit einem Luftaustauschsystem. Geht man von einer homogenen Verteilung von CO2 aus, hängt die CO2‑Konzentration in einer derartigen Umgebung vorrangig von zwei Faktoren ab: der CO2‑Menge, die vom Probanden produziert wird (V˙CO2), und dem Flow von Frischgas, der der Umgebung zugeführt wird (
- Der Helm ist eine geschlossene, aber belüftete Umgebung.
- CO2 im Helm ist unvermeidlich.
- Das Helmvolumen wirkt sich nicht auf die Rückatmung aus.
- PCO2h (der CO2‑Partialdruck im Helm) hängt nicht von der Helmgröße ab.
- Ein kontinuierlicher hoher Flow (Minutenvolumen) beschränkt die CO2‑Rückatmung.
- Bei einem Gesamtminutenvolumen von etwa 30 l/min sollte die CO2‑Rückatmung innerhalb der zulässigen Grenzwerte liegen.
- Ein ergänzender Flow (sofern möglich) und die (beabsichtigten kleinen) Leckagen tragen zur Reduzierung des PCO2h bei.
Hinweis!
Obwohl wir bei der Zusammenstellung der in diesem Handbuch enthaltenen Informationen größtmögliche Sorgfalt haben walten lassen, stellen diese keine offizielle Empfehlung durch das Unternehmen Hamilton Medical dar und können nicht an die Stelle der Meinung, Beurteilung und Anweisungen des ausgebildeten medizinischen Fachpersonals treten.
Vollständige Referenzen unten: (

Kostenloses eBook
Gut zu wissen! Die Grundlagen der nichtinvasiven Positivdruckbeatmung
Erhalten Sie einen Überblick über die Vorteile und klinische Relevanz der nichtinvasiven Beatmung sowie praktische Informationen zur Auswahl der richtigen Interface, Anpassung der Einstellungen und Überwachung des Patienten.
Fußnoten
Referenzen
- 1. Racca F, Appendini L, Gregoretti C, et al. Effectiveness of mask and helmet interfaces to deliver noninvasive ventilation in a human model of resistive breathing. J Appl Physiol (1985). 2005;99(4):1262‑1271. doi:10.1152/japplphysiol.01363.2004
- 2. Mojoli F, Iotti GA, Gerletti M, Lucarini C, Braschi A. Carbon dioxide rebreathing during non‑invasive ventilation delivered by helmet: a bench study. Intensive Care Med. 2008;34(8):1454‑1460. doi:10.1007/s00134‑008‑1109‑5
- 3. Racca F, Appendini L, Gregoretti C, et al. Helmet ventilation and carbon dioxide rebreathing: effects of adding a leak at the helmet ports. Intensive Care Med. 2008;34(8):1461‑1468. doi:10.1007/s00134‑008‑1120‑x
- 4. Patel BK, Wolfe KS, Pohlman AS, Hall JB, Kress JP. Effect of Noninvasive Ventilation Delivered by Helmet vs Face Mask on the Rate of Endotracheal Intubation in Patients With Acute Respiratory Distress Syndrome: A Randomized Clinical Trial. JAMA. 2016;315(22):2435‑2441. doi:10.1001/jama.2016.6338
- 5. Mojoli F, Iotti GA, Currò I, et al. An optimized set‑up for helmet noninvasive ventilation improves pressure support delivery and patient‑ventilator interaction. Intensive Care Med. 2013;39(1):38‑44. doi:10.1007/s00134‑012‑2686‑x
- 6. Taccone P, Hess D, Caironi P, Bigatello LM. Continuous positive airway pressure delivered with a "helmet": effects on carbon dioxide rebreathing. Crit Care Med. 2004;32(10):2090‑2096. doi:10.1097/01.ccm.0000142577.63316.c0
- 7. Vargas F, Thille A, Lyazidi A, Campo FR, Brochard L. Helmet with specific settings versus facemask for noninvasive ventilation. Crit Care Med. 2009;37(6):1921‑1928. doi:10.1097/CCM.0b013e31819fff93
- 8. Guan L, Zhou L, Zhang J, Peng W, Chen R. More awareness is needed for severe acute respiratory syndrome coronavirus 2019 transmission through exhaled air during non‑invasive respiratory support: experience from China. Eur Respir J. 2020;55(3):2000352. Published 2020 Mar 20. doi:10.1183/13993003.00352‑2020
- 9. Antonelli M, Conti G, Pelosi P, et al. New treatment of acute hypoxemic respiratory failure: noninvasive pressure support ventilation delivered by helmet‑‑a pilot controlled trial. Crit Care Med. 2002;30(3):602‑608. doi:10.1097/00003246‑200203000‑00019
- 10. Navalesi P, Costa R, Ceriana P, et al. Non‑invasive ventilation in chronic obstructive pulmonary disease patients: helmet versus facial mask. Intensive Care Med. 2007;33(1):74‑81. doi:10.1007/s00134‑006‑0391‑3
- 11. Moerer O, Herrmann P, Hinz J, et al. High flow biphasic positive airway pressure by helmet‑‑effects on pressurization, tidal volume, carbon dioxide accumulation and noise exposure. Crit Care. 2009;13(3):R85. doi:10.1186/cc7907
- 12. Hui DS, Chow BK, Lo T, et al. Exhaled air dispersion during noninvasive ventilation via helmets and a total facemask. Chest. 2015;147(5):1336‑1343. doi:10.1378/chest.14‑1934
- 13. Alhazzani W, Møller MH, Arabi YM, et al. Surviving Sepsis Campaign: guidelines on the management of critically ill adults with Coronavirus Disease 2019 (COVID‑19). Intensive Care Med. 2020;46(5):854‑887. doi:10.1007/s00134‑020‑06022‑5